Stem cell therapy and regenerative medicine has grown significantly over the last decade. More and more sources of cellular products and protocols are being developed daily. At MetroMD we have continued to seek and provide our patients with the latest in cellular products and treatments. Originally, we worked with bone marrow derived stem cells for orthopedic related issues, then we developed our PRP practice focusing on joint injuries and aesthetics. However, we know have a new source of stem cells and that is from an “off the shelf” product called Umbilical Cord cells that are derived from Wharton’s Jelly.
Wharton’s Jelly is a great source for regenerative tissues, cells and factors. This area of the cord has a large concentration of mesenchymal stem cells (MSCs), growth factors, collagen, progenitor cells and other regenerative factors that can be used in the healing process. We have the ability to now harvest these tissues for homologous use in a patient.
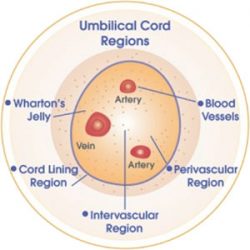
Whartons Jelly Stem Cell Los Angeles
These cells are extracted in a FDA regulated tissue bank where each donor and donated tissue goes through a thorough screening process. Each aliquot is screened for genetic mutations, disease, and other factors to ensure safety for transplant purposes. Once these cells are screened and isolated they are cryofrozen to preserve the cells for later use.
These cells are then sent to our facility where we also keep them cryofrozen in a cryofreezer in our licensed lab at MetroMD. These cells are kept frozen and viable until use with a patient. Our physicians then work with our patients to pair correct treatment types with these cells to ensure the patient has a homologous treatment and can help aid in healing.
These cord derived cells from Whartons Jelly have effects to regulate the immune system in the body, promote proliferation of native cells as well as with the use of the MSC cell types the ability to differentiate into cell types needed. This source of cells and regenerative factors truly is the Gold Standard today in terms of cellular medicine treatments.


